  
There are various concepts in thermodynamics, some of these concepts are explained as follows: Thermodynamic SystemsĪ collection of an extremely large number of atoms or molecules confined within certain boundaries such that it has certain values of pressure (P), volume (V) and temperature (T) is called a thermodynamic system.Īnything outside the thermodynamic system to which energy or matter is exchanged is called its surroundings. More general notions than those covered by equilibrium thermodynamics are needed for the thermodynamic study of non-equilibrium systems. The majority of systems in nature are not in thermodynamic equilibrium because they are not in stationary states and are subject to fluxes of matter and energy to and from other systems on a continual and irregular basis. Systems that are not in thermodynamic equilibrium are the focus of the field of thermodynamics known as non-equilibrium thermodynamics. Unbalanced potentials or driving forces between the system’s macroscopically diverse components do not exist in an equilibrium state. In the case of the most basic systems or bodies, this means that their intensive properties are uniform and that their pressures are perpendicular to their boundaries. The phrase “thermodynamic equilibrium” refers to a condition of equilibrium in which all macroscopic flows are zero. Equilibrium ThermodynamicsĮquilibrium thermodynamics is the study of matter and energy transfers in systems or substances that can be moved from one state of thermodynamic equilibrium to another by agents in their environment. Determining the spontaneity of a certain transition is the main goal of chemical thermodynamics. Chemical ThermodynamicsĬhemical thermodynamics is the study of how energy interacts with chemical processes or state changes in accordance with the laws of thermodynamics. It does this by connecting the microscopic, bulk properties of materials that can be observed on the human scale to the macroscopic, individual atom, and molecule properties. This field explains classical thermodynamics as a natural consequence of statistics, classical mechanics, and quantum theory at the microscopic level. 
The development of atomic and molecular theories in the late 19th and early 20th centuries gave rise to statistical mechanics, also known as statistical thermodynamics, which added an interpretation of the microscopic interactions between individual particles or quantum-mechanical states to classical thermodynamics. In order to determine the characteristics and predict the characteristics of the matter conducting the process, individuals take into account units like temperature and pressure. The behaviour of matter is examined using a macroscopic perspective in classical thermodynamics. 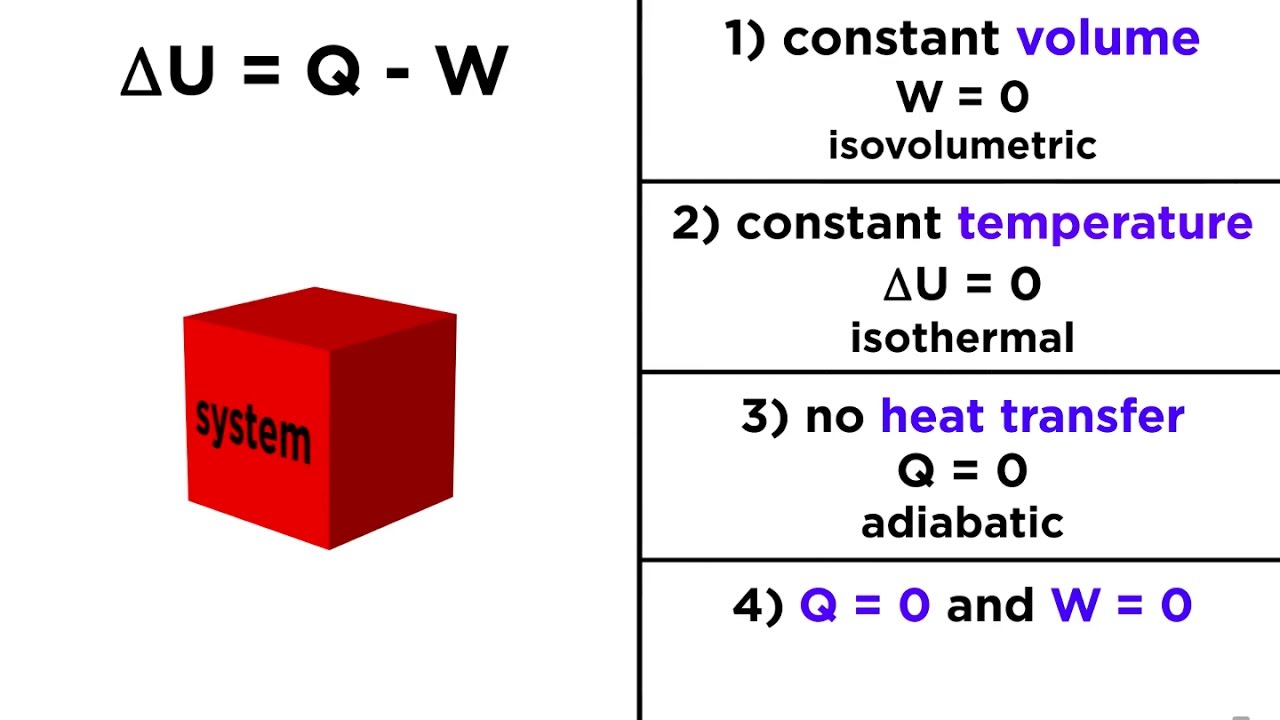
Let’s learn about these in detail in the article. The study of Thermodynamics is classified into several branches listed below: Thermodynamics is a branch of Physics that deals with the concept of energy, heat and temperature and their interrelation with radiation, energy and physical characteristics of matter. The word “universe” refers to both the surroundings and the system. Once the boundary diagram has been created, the flow across system boundaries can be used to describe the movement and transfer of energy. The “system” refers to everything that is contained within the boundary, and the “surroundings” refers to all that is outside of it. Role of Mahatma Gandhi in Freedom StruggleĬreating a clear boundary makes thermodynamics much simpler.This is the typical heat capacity of water. For example, say that we want to reduce the sample's thermal energy by 63,000 J. If you want to cool down the sample, insert the subtracted energy as a negative value. Insert the amount of energy supplied as a positive value. Determine whether you want to warm up the sample (give it some thermal energy) or cool it down (take some thermal energy away). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
